 As one electron goes to s orbital, three occupy the p orbital, and the last one enters the d orbitals of the central atom, the hybridization of Pbr5 is sp3d. The last valence electron occupies the dx orbital. The first valence electron of the halogen occupies the s orbital next three valence electrons go in px, py and pz orbital. As the Phosphorus forms bonds with five Bromine molecules, all the electrons enter in different orbitals of the shell. PBr5 has Phosphorous as the central atom has eight electrons in its outer shell after forming the bond with neighbouring halogen atoms. It is easy to understand the hybridization of the molecule after knowing its Lewis structure. In chemistry, the term hybridization refers to combining two or more orbitals with different energies in a compound to give a hybrid orbital. So the total number of lone electrons is 30 now. But there are still three pairs of lone electron for each Bromine molecule. So there are no lone pairs of electrons on the central atom. As the P molecule has five valence electrons, all of these electrons from the bond with one valence electron of each Bromine molecule. Here the molecule of Phosphorus in the centre that bonds with the rest five molecules of Bromine. of valence electrons for Phosphorus + No. So the total number of valence electrons is, Here in this compound, there is one molecule of Phosphorus having five valence electrons and five molecules of Bromine having seven valence electrons each. The lewis structure follows the octet rule that states that for a molecule to be stable, which states that there should be eight electrons in the outer shell of the atom for a molecule to be stable. All these bonding, as well as non-bonding electrons, are known as Valence electrons. 
The straight lines represent the bonds in the compound, and the dots represent the lone pair of electrons. Whereas the electrons that do not constitute any bonds are known as non-bonding electrons or lone electrons. 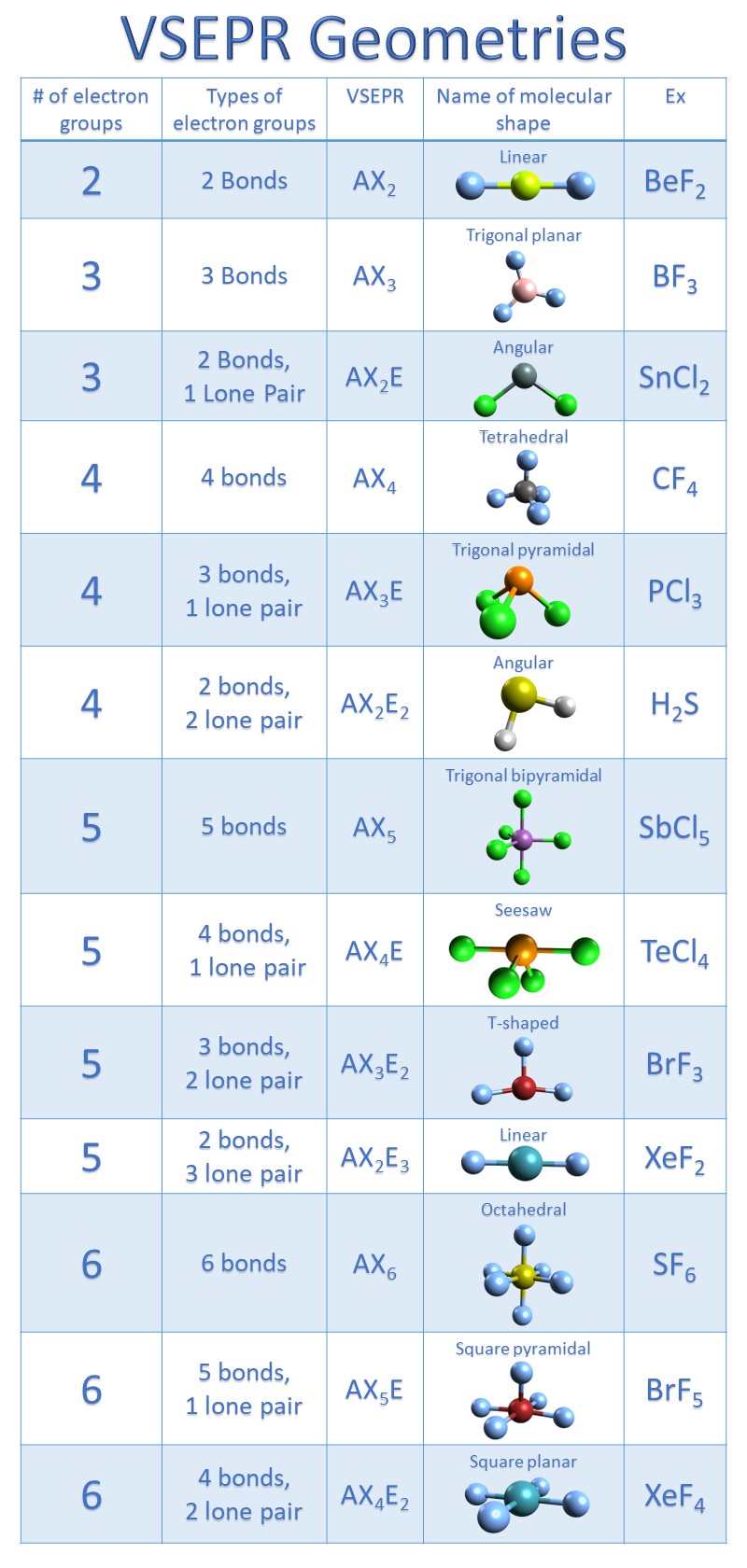
The electrons that participate in forming the bond are known as the bonding pair of electrons. It also helps in knowing the type of charges present on the compound. This representation helps to understand the basic structure of the compound. The lewis structure is defined as the pictorial representation of the electron participating in bond formation to form the compound. Studying the Lewis structure of a given compound helps in understanding other chemical properties of the compound such as hybridisation, polarity, etc easily. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
